Inquire
Ethylene Sulfate: Uses, Structure & Properties
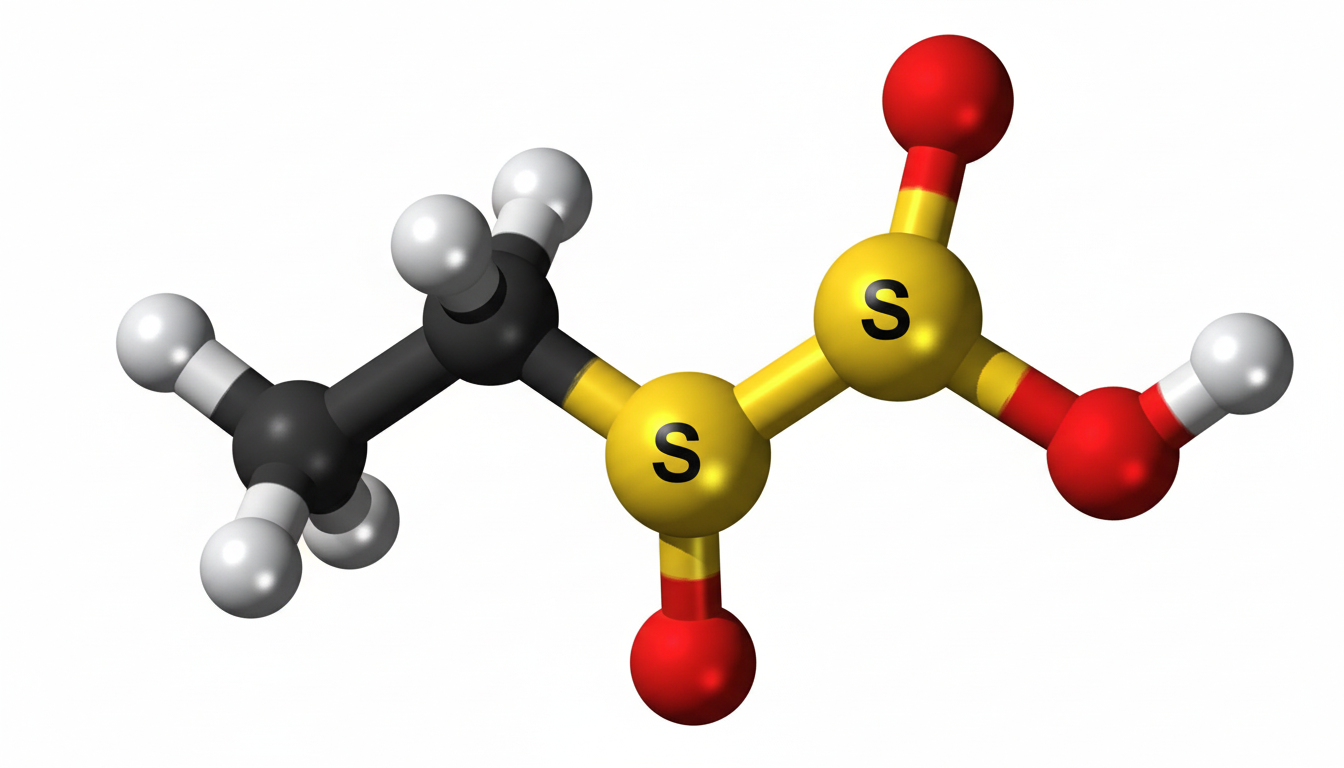
Ethylene sulfate is an organic chemical compound that plays an important role in industrial chemistry and advanced material applications. It is widely studied due to its reactive nature and usefulness as an intermediate in chemical synthesis. In modern industries, ethylene sulfate has gained attention especially in electrochemistry and organic manufacturing processes. Understanding its structure, properties, production methods, and applications is essential for researchers, chemists, and industrial professionals.
What is Ethylene Sulfate
Ethylene sulfate is a cyclic sulfate ester derived from ethylene glycol. It contains a reactive sulfate group bonded within a small ring structure, which makes it highly useful in chemical reactions. This compound is typically used as an intermediate rather than a final product due to its high reactivity. In chemical terms, it is known for participating in ring-opening reactions, which makes it valuable in synthesis processes for more complex molecules.
The compound is generally stable under controlled conditions but can react quickly when exposed to moisture, heat, or nucleophilic substances. This reactivity is what gives ethylene sulfate its industrial importance.
Chemical Structure and Properties
Ethylene sulfate has a cyclic structure that includes two carbon atoms and a sulfate group forming a ring. This strained ring structure contributes to its high reactivity. The molecule is polar in nature, which allows it to dissolve in certain organic solvents and participate in various chemical reactions.
One of the most important properties of ethylene sulfate is its ability to act as an electrophile. This means it readily reacts with electron-rich species. Because of this characteristic, it is commonly used in organic synthesis and electrochemical applications. It has a relatively high boiling point compared to simple organic compounds, but it decomposes before reaching extreme temperatures.
Its chemical stability depends heavily on environmental conditions. In dry and controlled environments, it remains stable, but in the presence of water, it can hydrolyze and form other compounds. This sensitivity requires careful handling in laboratory and industrial settings.
Synthesis of Ethylene Sulfate
Ethylene sulfate is typically synthesized through the reaction of ethylene oxide or ethylene glycol derivatives with sulfur trioxide or other sulfonating agents. The process involves controlled conditions to ensure the formation of the cyclic sulfate structure.
During synthesis, temperature and reaction environment are carefully controlled to avoid unwanted side reactions. The formation of the cyclic ester ring is a key step, and it requires precise chemical balancing. Industrial production methods focus on maximizing yield while maintaining safety due to the reactive nature of the chemicals involved.
Purification of ethylene sulfate is also an important step, as impurities can affect its performance in downstream applications. Techniques such as distillation under reduced pressure or crystallization are sometimes used depending on the production scale.
Physical and Chemical Behavior
Ethylene sulfate exhibits strong chemical reactivity due to ring strain in its molecular structure. This makes it highly useful in reactions that require ring-opening mechanisms. It reacts readily with nucleophiles such as alcohols, amines, and water under specific conditions.
Physically, it is usually a colorless to pale liquid or solid depending on temperature and purity. Its solubility characteristics depend on the solvent system used, but it generally shows better compatibility with organic solvents than with water.
In electrochemical environments, ethylene sulfate is known to improve the formation of stable layers on electrode surfaces, which is why it is studied in battery technologies.
Industrial Applications
Ethylene sulfate is used as a key intermediate in several industrial chemical processes. One of its most important roles is in organic synthesis, where it helps create more complex chemical structures through controlled reactions.
In the field of energy storage, ethylene sulfate is used in research related to lithium-ion batteries. It contributes to the formation of stable solid electrolyte interfaces, which improve battery life and performance. This makes it highly valuable in advanced battery electrolyte formulations.
It is also used in specialty chemical manufacturing, where precise molecular modifications are required. Its reactivity allows chemists to introduce sulfate groups into organic molecules efficiently, making it useful in pharmaceuticals and material science research.
Use in Electrolyte and Battery Technology
One of the most significant modern uses of ethylene sulfate is in electrochemical systems, especially lithium-ion batteries. It is studied as an additive in electrolytes because it helps form a stable protective layer on battery electrodes.
This layer improves cycling stability and reduces degradation during charging and discharging cycles. As a result, batteries can last longer and perform more efficiently. Researchers continue to explore how ethylene sulfate can improve energy density and safety in next-generation battery systems.
Its role in battery chemistry highlights its importance in renewable energy storage and electric vehicle technology, where performance and reliability are critical.
Safety and Handling
Ethylene sulfate must be handled with care due to its reactive nature. It can cause irritation upon contact with skin, eyes, or respiratory systems. Proper protective equipment such as gloves, goggles, and lab coats is necessary when working with this compound.
In industrial environments, strict safety protocols are followed to prevent accidental exposure or decomposition. It should be stored in a dry, cool place away from incompatible substances such as strong nucleophiles or water.
Ventilation is also important when handling ethylene sulfate to avoid inhalation of any vapors or reaction byproducts. Safety data sheets are typically followed to ensure proper handling procedures.
Environmental Considerations
The environmental impact of ethylene sulfate depends on how it is managed and disposed of. If released improperly, it can react with moisture and contribute to chemical contamination. Therefore, waste handling procedures are essential in industrial use.
Controlled degradation and neutralization methods are used to minimize environmental risks. Regulatory guidelines in chemical industries ensure that compounds like ethylene sulfate are handled responsibly to reduce ecological impact.
Research is ongoing to better understand its long-term environmental behavior, especially in large-scale industrial applications.
Storage and Stability
Ethylene sulfate requires careful storage conditions to maintain stability. It should be kept in tightly sealed containers to prevent exposure to moisture and air. Even small amounts of water can trigger hydrolysis reactions.
Temperature control is also important, as excessive heat can lead to decomposition. Industrial storage facilities typically use dry and temperature-regulated environments for such reactive chemicals.
Proper labeling and segregation from incompatible chemicals are also essential to ensure safety and stability during storage.
Conclusion
Ethylene sulfate is a highly important chemical intermediate with wide applications in organic synthesis, industrial chemistry, and modern battery technology. Its unique cyclic structure and high reactivity make it valuable for controlled chemical reactions. At the same time, its reactive nature requires careful handling, storage, and safety measures.
With ongoing research, especially in energy storage systems, ethylene sulfate continues to gain importance in advanced technology fields. Its role in improving battery performance and supporting complex chemical synthesis ensures that it remains a significant compound in both academic and industrial chemistry.
- Managerial Effectiveness!
- Future and Predictions
- Motivatinal / Inspiring
- Fitness and Wellness
- Medical & Health
- Manufacturing
- Education
- Real-Estate
- Food Industry
- Hospitality
- Online Games
- Sports
- Home Services
- Civil Engineering
- Safety and Protection
- Software Products & Services
- Fashion and Jewellery
- Artificial Intelligence
- Entrepreneurship
- Mentoring & Guidance
- Marketing
- Networking
- HR & Recruiting
- Literature
- Shopping
- Career Management & Advancement


 SkillClick
SkillClick